Before administering a dose of Mounjaro.
Read the instructions for use in the pack before using the KwikPen. The step numbers referred to below, correspond to the same numbered steps in the instructions for use.
The administration of a dose of tirzepatide includes:
- Preparing
- Priming
- Injecting
- After the injection
Preparation.
The supplies needed for administration include:
- Mounjaro KwikPen
- KwikPen compatible needle
- Swab
- Gauze or cotton ball
- Sharps disposal container or household container.
Prepare the pen as described in the instructions for use:
Prepare the pen as described in the instructions for use:
Pull the pen cap straight off and inspect your KwikPen, the label and the medication in the pen.
- Confirm the medication name and strength.
- Make sure the Kwikpen is NOT expired or damaged.
- Make sure the medication has not been frozen, is colourless to slightly yellow and free of particles.
Wipe the red inner seal with a swab.
Select a new pen needle and remove the paper tab.
Attach the new needle, by pushing the capped pen needle straight onto the pen and twisting it on until it’s tight.
Pull off the outer needle shield and keep it. Pull off the inner needle shield and discard it.
Important
The KwikPen must NOT be used, if it looks damaged, or the medicine is expired, cloudy, or coloured. For full information please refer to the Instructions for Use.
Priming
Slowly turn the dose knob until 2 clicks are heard and the extended line is shown in the dose window. This is the prime position. It can be corrected by turning the dose knob in either direction until the prime position lines up with the dose indicator.
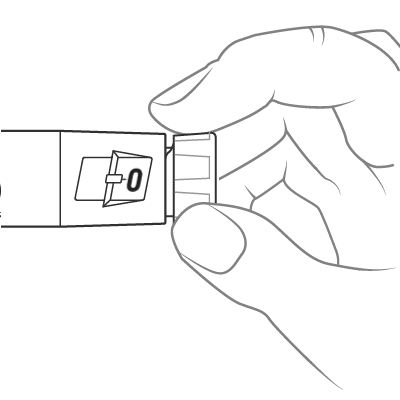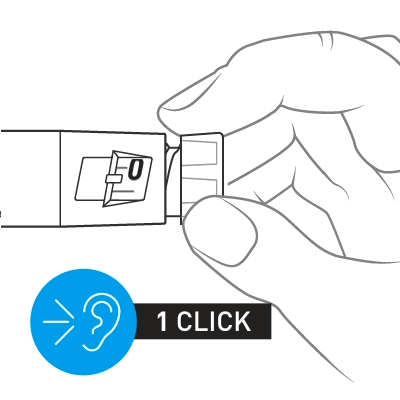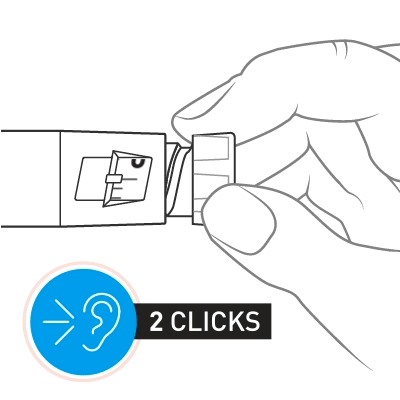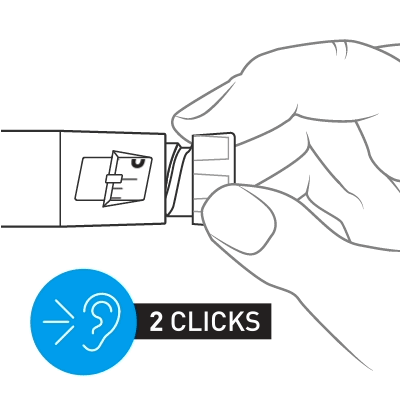
Hold the pen with the needle pointing up. Tap the cartridge holder gently to collect air bubbles at the top.

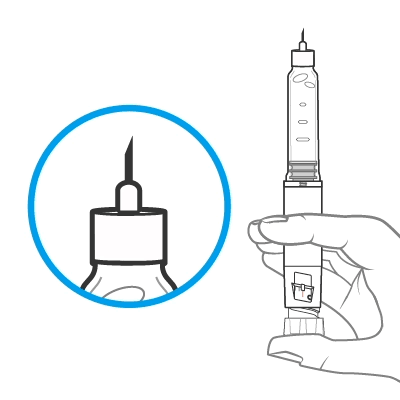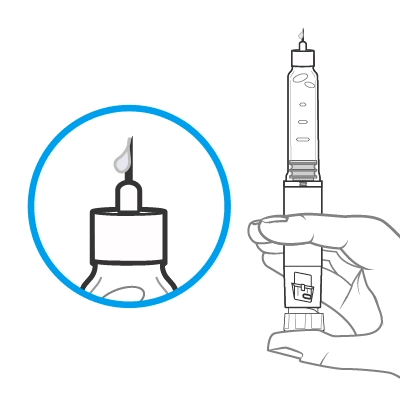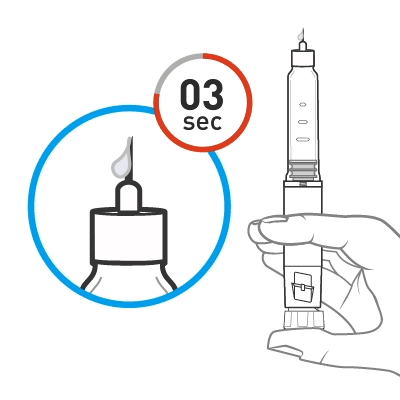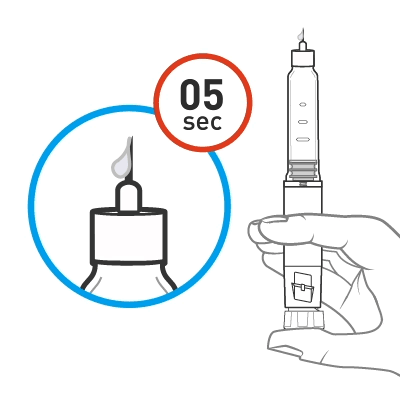
Release some medicine into the air by pushing the dose knob in until it stops and then slowly count to 5 while holding the dose knob. Check the dose window. The “0” must be seen in the dose window. DO NOT inject into the body.
Important
If no medicine is seen at the tip of the needle, please refer to Step 9 in the Instructions for Use for further information.
Injecting
Choose an injection site.
- Suitable injection sites are thigh and abdomen (at least 5 centimetres from the belly button).
- An injection in the back of the upper arm is possible, when another person gives the injection.
- Rotate the injection site each week.
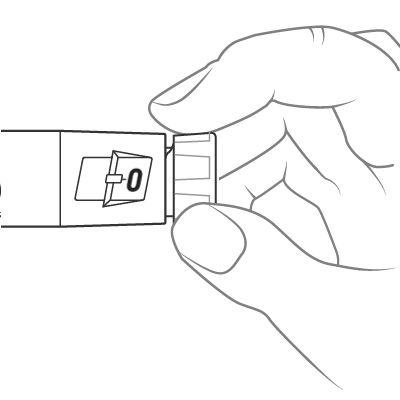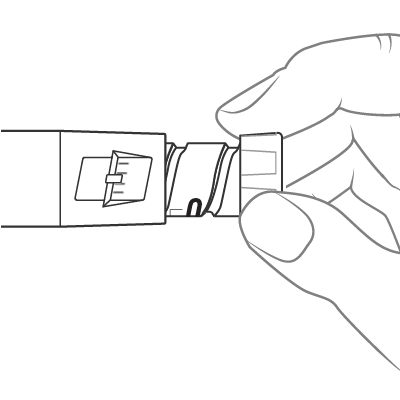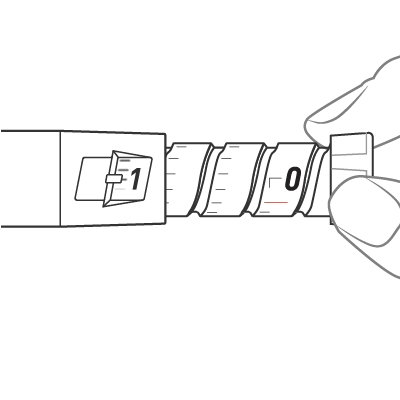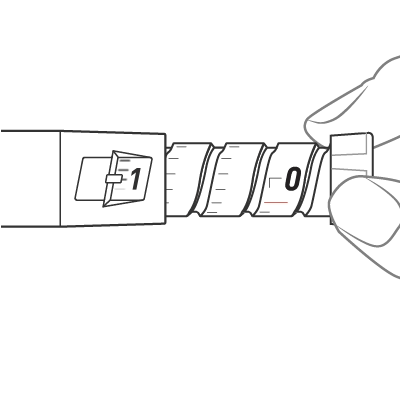
To dial the dose, turn the dose knob until it stops and the “1” is seen in the dose window, as indicated by the icon. This is equal to a full 0.6 mL dose.
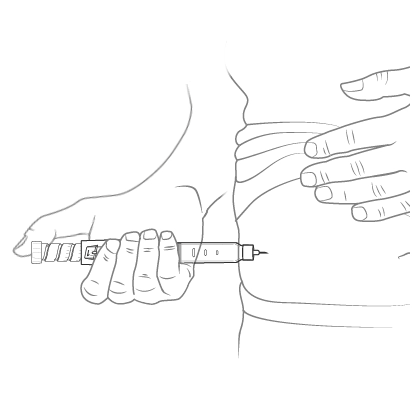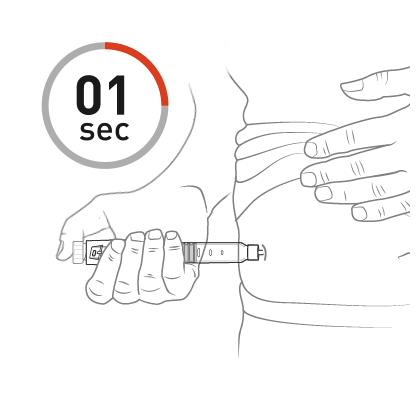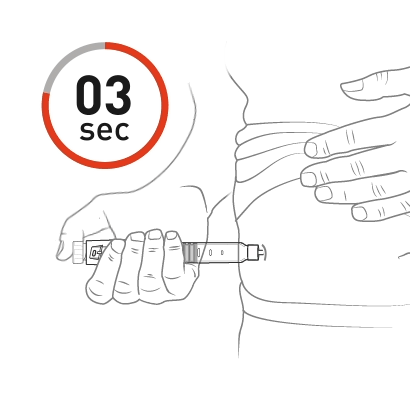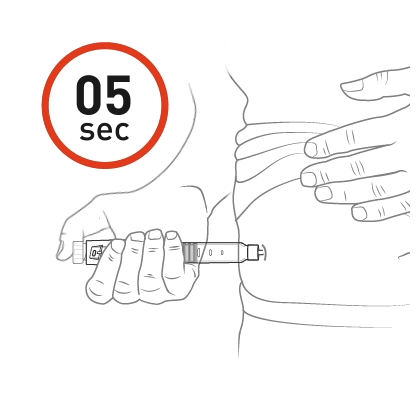
Insert the needle into the skin and push the dose knob to inject the medicine.
Push the dose knob until it stops and slowly count to 5 while holding the dose knob.
Important
Check the Dose window and see “0”, indicated by the icon. This is the confirmation that the full dose has been administered. If you do not see the “0” in the dose window, you should insert the needle back into the skin and finish the injection. You should NOT redial a dose. For further information please refer to step 13 in the Instructions for Use.
After The Injection
After injection, you should:
Lightly press the injection site with gauze or a cotton ball if you see blood, and not rub the injection site.
Carefully replace the outer needle shield.
Unscrew the needle and put it in a sharps container. and not store the pen with the needle attached.
Replace the pen cap and NOT store the pen without the pen cap attached.
How to store the used pen:
- At room temperature up to 30°C after the injection.
- Keep away from heat.
- Keep the pen and needles out of the sight and reach of children.
How to dispose of the used pen:
- Remove the needle in order to dispose of the pen
- Dispose of the pen in the household bin or sharps disposal container.
When to dispose of the pen:
- 30 days after first use.
- After administration of 4 doses (1 dose per week).
- When the pen is expired or looks damaged
- When the medication has been frozen, has particles, is cloudy, or is discoloured. Mounjaro should be colourless to slightly yellow.
References
- 1.
Mounjaro [instructions for use]. Eli Lilly Nederland B.V., The Netherlands. (GB)
- 2.
Mounjaro [summary of product characteristics]. Eli Lilly Nederland B.V., The Netherlands. (GB)
This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse reactions.
Last reviewed: 16th February 2025